Recovery Planning
Planning is a central element of conservation. This is where social values and scientific knowledge come together to inform decisions about what we will do and where we will do it. It is here that the broad goal of maintaining biodiversity is translated into operational terms.
In this chapter, we will focus on planning as it pertains to the management of species at risk in Canada. The emphasis will be on the mechanics of the process and the practical challenges inherent in moving from policy to practice. We will touch on planning again in Chapter 7, in the context of integrated landscape management, and in Chapter 10, in the context of structured decision making. Chapter 11 provides some practical examples of species recovery planning.
As recounted in Chapter 3, the federal and provincial governments share responsibility for managing species at risk. Although SARA is national in scope, it only applies directly to areas of federal jurisdiction, including the territories, oceans, national parks, some freshwaters, and most migratory birds. The primary responsibility for managing terrestrial species falls to the provincial governments, under provincial species at risk legislation and other wildlife laws. In practice, the federal and provincial governments coordinate their efforts, albeit somewhat uneasily. Provincial cooperation is motivated by the fact that SARA includes “safety net” provisions that come into effect if a province fails to meet the protection standard prescribed by SARA (GOC 2002, Sec. 80.4). The following discussion of species at risk management is based on the federal process.
Overview of SARA
Under SARA, the key steps in the recovery process, as well as timelines, are explicitly defined in law, distinguishing SARA from most other Canadian conservation legislation. The process begins with an assessment by the independent Committee on the Status of Endangered Wildlife in Canada (COSEWIC). COSEWIC commissions status reports on potentially at-risk species and then provides a status recommendation to the federal government, patterned on an international framework (Mace et al. 2008). The categories are: extinct, extirpated, endangered, threatened, of special concern, and not at risk. The government has nine months to either accept a recommendation (i.e., to “list” it), decline it with cause, or return the issue to COSEWIC for further clarification (GOC 2002, Sec. 27.1).
The intent of this two-step listing process is to separate the scientific and political aspects of conservation decision making. COSEWIC’s assessments are to be based strictly on the biology of the species, leaving the consideration of feasibility, cost, and social and political ramifications to elected officials (ECCC 2016a).
Species that are subject to commercial harvest and Indigenous harvest (particularly in Nunavut) have often been denied listing on socio-economic grounds (Findlay et al. 2009). Marine fish have been the most affected. A review of COSEWIC listing recommendations for these species found that 71% were denied (McDevitt-Irwin et al. 2015). According to Schultz et al. (2013), the bias against marine species is not just related to economic impacts but also reflects the fact that ocean wildlife is under federal jurisdiction (i.e., the implications of listing hit closer to home). Such systematic circumvention of SARA is contrary to what was intended by lawmakers when they passed the Act.
Once a species is listed in the SARA registry, a recovery strategy must be prepared within one year if the species is endangered, and within two years if the species is threatened (GOC 2002, Sec. 42.1). For species of special concern, a management plan is required within three years, and the emphasis is on preventing further declines rather than on recovery. The planning process is overseen by the relevant federal agency: Fisheries and Oceans Canada for aquatic species, Environment and Climate Change Canada for terrestrial species, and Parks Canada for species within national parks. Strategies are developed by species-specific recovery teams composed of government staff and external experts.
Recovery strategies are largely science based, drawing on published reports and the knowledge of domain experts. Strategies follow a standard format that includes the following elements:
- Species biology, including distribution, population size and trends, habitat needs, and limiting factors
- Threat analysis, with at least a qualitative ranking of importance
- Assessment of whether recovery is feasible, accounting for reproductive capacity, habitat availability, potential for threat mitigation, and existence of practical recovery techniques
- Recovery goal and objectives
- Recovery approaches
- Identification of critical habitat
- Knowledge gaps and priorities for future research
After a recovery strategy is completed, the federal government must develop an action plan, in consultation with stakeholders. Action plans define the management steps that will be taken to achieve the objectives outlined in the recovery strategy (GOC 2002, Sec. 49). It is at this stage that socio-economic factors are considered, rather than in the recovery strategy. The government must monitor the action plan and report on progress toward meeting recovery objectives five years after the plan comes into effect (GOC 2002, Sec. 55).
Recovery planning is subject to several limitations (SPI 2018). Species at risk are notoriously difficult to study, and funding for research is limited. Species like woodland caribou and killer whales, which have been studied intensively, are the exceptions rather than the rule. For most species at risk, recovery teams have only basic information on distribution and threats to work with, and little or no tactical modelling.
There are also process-related problems to contend with. As previously noted, many species are denied listing and never even enter the planning process (McDevitt-Irwin et al. 2015). For listed species, planning is frequently subject to delays at both the recovery strategy stage and the action planning stage (AGC 2013). Moreover, most recovery strategies to date have not identified critical habitat as required (Bird and Hodges 2017). Matters have improved in recent years, following several successful court challenges (FCC 2014). However, none of the process deficiencies have been fully remedied.
Finally, the mechanisms for plan implementation are poorly developed and many plans languish at this stage. SARA acknowledges the need to consider other social objectives but does not describe how trade-off decisions are to be made. Implementation is also hindered by the split between federal and provincial responsibilities (i.e., federal planning and provincial implementation).
No unifying framework exists for managing focal species that are not listed as species at risk. For harvested species, policies and plans are usually developed by fish and wildlife departments and forestry departments at the regional or provincial scale. These plans reflect local interests and priorities. In some cases, the aim is to maintain historical population levels, as described by NRV. More often, the objective involves some combination of ecological and social outcomes, taking the costs and benefits of management actions into account. Species that are not harvested generally do not have formal management plans and are instead managed through landscape-scale conservation approaches.
Incorporating Genetic Diversity
Different geographic regions exert different selective pressures. Consequently, wide-ranging species often exhibit local genetic adaptations. These adaptations are not always obvious but can be demonstrated through transplant experiments. For example, aspen from Minnesota exhibit almost twice the biomass growth of local aspen when transplanted to Alberta (Schreiber et al. 2013). In turn, Alberta aspen exhibit traits, such as late bud break, that are protective against exceptional spring frosts that occur sporadically in Alberta but not in Minnesota (Li et al. 2010).
The development of local adaptations is counteracted, to a variable degree, by dispersal, which tends to homogenize populations (Fraser et al. 2011). The development of local adaptations also depends on how much environmental variability a species experiences. Consequently, the degree of spatial structuring varies among species. Some species feature genetically distinct populations with low rates of genetic mixing, whereas other species exhibit genetic homogeneity over large geographic regions (Laikre et al. 2005). Intermediate forms, like aspen, may exhibit continuous genetic change over geographic distance (Fig. 6.14).
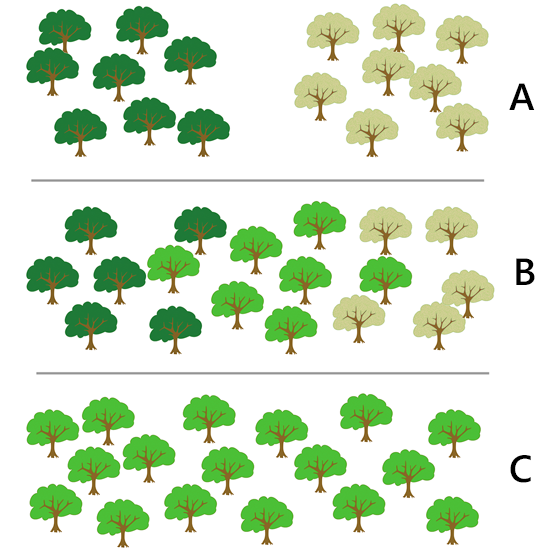 |
Fig. 6.14. The genetic structure of populations can be crudely classified into three basic types. A) Distinct populations, where gene flow is low enough for genetic divergence. B) Continuous change, where genetic composition changes continuously over space, resulting in isolation by distance. C) No differentiation, where gene flow is so extensive that genetic homogeneity prevails across the entire region. Adapted from Laikre et al. 2005. Clipart by X. Dengra. |
Local genetic adaptations are important to conservation because they maximize the fitness of individual populations and they provide genetic variability for adapting to change at the species level (Lande and Shannon 1996). The challenge conservationists face is determining the appropriate genetic unit for protection (Waples 1998). The basic objective is to identify subsets of a species that have genetic attributes important to overall species viability. These are referred to as “evolutionary significant units” (Ryder 1986). The difficulty is in establishing the link between genetics and species viability and in determining the appropriate level of importance (Waples 1998).
In Canada, COSEWIC uses a set of guidelines (Green 2005) to identify so-called “designatable units” below the species level, on a case-by-case basis. The main criteria include disjunct range, occupancy of different ecoregions, and genetic distinctiveness. In each case, differentiation is made only if the individual units also differ in conservation status. If the units share the same status, there is no practical value in distinguishing them because they will receive the same management treatment.
As with any such system, COSEWIC’s approach to selecting designatable units has shortcomings. Genetic sampling across broad regions is difficult and expensive, even with indirect sampling techniques (e.g., using hair and scat samples). As a result, only a relatively small number of species have been well studied. Moreover, the relationship between genetic variability and fitness is poorly understood, and quantitative analysis is generally not possible (Green 2005). In practice, COSEWIC bases its decisions on expert opinion, and most designatable units are defined very coarsely (e.g., broad regional populations such as “eastern” and “boreal”).
There are no formal mechanisms or requirements under SARA for maintaining genetic diversity below the level of designated units. Therefore, the consideration of fine-scale genetic structure in recovery plans is quite variable. For species reduced to very small populations, the maintenance of all remaining genetic variance and the avoidance of inbreeding are often identified as major management concerns, particularly when artificial breeding programs are used. For other species, genetics may be addressed indirectly, through recovery objectives that seek to maintain the species across its entire range. Some recovery strategies make no reference to genetics at all.
For focal species not listed under SARA, the consideration of genetics is generally quite limited, with certain exceptions. For example, replanting programs (e.g., commercial trees) and restocking programs (e.g., sport fish) often include measures to ensure that local genotypes are maintained. In addition, management plans for species with highly fragmented populations may include measures to avoid inbreeding. For most other species, genetic considerations are handled indirectly through distribution objectives, if at all.
Setting Objectives
The articulation of objectives is central to the planning process—it is what motivates and directs management action. For species at risk applications, objective setting must account for differences in the status of individual species. What needs to be done, and what is feasible to do, is much different for a species on the precipice of extinction as compared to a widely distributed species that is undergoing a slow decline. SARA defines a hierarchical objective composed of three elements: (1) to prevent wildlife species from becoming extinct, (2) to provide for the recovery of wildlife species that are threatened as a result of human activity, and (3) to manage species of special concern to prevent them from becoming threatened (GOC 2002, Sec. 6).
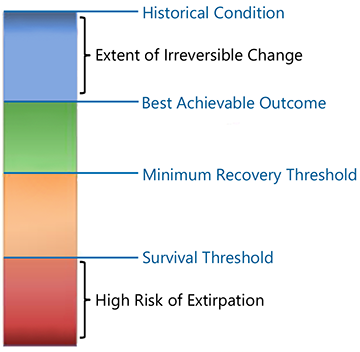
Under SARA, recovery is a multistage process (Fig. 6.15). For species on the edge of extinction, the emphasis is on ensuring survival in the immediate future. Addressing the compound risks associated with small population size is paramount. Once immediate survival is assured, the focus shifts to securing long-term recovery. The aim of recovery is to “return the species to whatever its natural condition was in Canada prior to being put at risk by human activities” (ECCC 2020). This entails additional population growth, range expansion, and the establishment of multiple distinct populations. The long-term recovery goal serves as a beacon, providing context and direction for the overall recovery process.
For many species, the historical condition is not achievable because of irreversible ecological changes. In such cases, the recovery endpoint is the best outcome that is biologically and technically achievable (Fig. 6.15). Socio-economic constraints also affect recovery outcomes, but they are addressed later in the planning process, not when setting biotic objectives (ECCC 2020).
Policymakers have not yet determined how to accommodate climate change when setting species recovery objectives. Using historical conditions as the reference state remains serviceable for the time being because climate-induced ecological changes are not yet widespread. But going forward, we will need to transition to a dynamic reference state that is robust to climate change. We will explore the options for doing so in Chapter 9.
Identifying Critical Habitat
SARA stipulates that recovery strategies must define critical habitat, but this has proven to be challenging (Bird and Hodges 2017). The process has suffered from protracted delays, non-compliance, and a lack of policy guidance (Martin et al. 2017). New policy has recently been developed to provide additional direction (ECCC 2016b), and compliance is now improving (Bird and Hodges 2017).
The process of defining critical habitat under SARA has three steps: (1) assemble and assess the available data; (2) identify critical habitat, including its geographic location and biophysical attributes; and (3) describe the specific threats to the identified habitat. It is acknowledged that data inadequacies will exist for most species at risk. SARA policy (ECCC 2016b) suggests that recovery teams should generally do the best they can with existing data:
The fact that there will be better and/or more information on which to base critical habitat identification at some point in the future cannot be used as a reason to delay identifying critical habitat to the extent possible, based on the best information available at this time. If all of the critical habitat cannot be identified based on the best available information, then critical habitat will be identified to the extent possible. … Critical habitat identification is often an iterative process and partial identification may be possible in advance of full identification. (pp. 12–16)
When identifying critical habitat, recovery teams are guided by SARA’s definition: “critical habitat means the habitat that is necessary for the survival or recovery of a listed wildlife species” (GOC 2002, Sec. 2). As previously noted, socio-economic implications are intended to be addressed at the action planning stage, rather than when critical habitat is being identified (ECCC 2016b).
In most recovery strategies to date, critical habitat has been identified on the basis of known use (Camaclang et al. 2015; Martin et al. 2017). For example, critical habitat for the loggerhead shrike (migrans subspecies) is identified as patches of suitable grassland occupied by a confirmed breeding pair during the period 1999–2008, along with any other suitable patches within 400 m of a nesting site (EC 2015). A shortcoming of this approach is that it provides no assurance that there will be sufficient habitat for species survival, let alone recovery (Camaclang et al. 2015; Martin et al. 2017). The survival of many species at risk requires populations to increase from critically low levels, with a concomitant need for habitat that is currently unoccupied. Habitat requirements for full recovery are even greater.
The upshot is that there still is a gap between policy and current practice. Critical habitat is being defined very narrowly (if at all) and generally supports only immediate needs rather than long-term survival and recovery. This may improve over time, as additional field studies and modelling enable the identification of critical habitat on the basis of population recovery needs rather than current use. But progress will be slow unless additional staff and research funding are brought to bear (Bird and Hodges 2017; Martin et al. 2017).
In the final step, recovery teams must characterize the threats to critical habitat, providing the basis for protection measures. Threats include all activities that may degrade critical habitat, either permanently or temporarily, “such that it would not serve its function when needed by the species” (ECCC 2016b, p. 22). Detailed assessments are needed because, in practice, protection measures most often involve the mitigation of specific threats rather than the establishment of formal protected areas.
For species of special concern and other focal species, there is no legal requirement for identifying critical habitat. Therefore, other mechanisms must be used to identify and protect the habitat of these species. This usually entails integrating focal species management objectives into broader land-use planning programs and protected area initiatives, which we will discuss in subsequent chapters.

