2.4 Economic Minerals
If you can’t grow it, you have to mine it
Anything we can’t grow we have to extract from Earth in one way or another. This includes water, of course, our most important resource, but it also includes all the other materials that we need to construct things like roads, dams, and bridges, or manufacture things like plates, toasters, and telephones.

Virtually everything we use every day is made from resources from Earth. For example, let’s look at a tablet computer (Figure 2.4.1). Most of the case is made of a plastic known as ABS, which is made from either gas or petroleum. Some tablets have a case made from aluminum. The glass of a touch screen is made mostly from quartz combined with smaller amounts of sodium oxide (Na2O), sodium carbonate (Na2CO3), and calcium oxide (CaO). To make it work as a touch screen, the upper surface is coated with indium tin oxide. When you touch the screen you’re actually pushing a thin layer of polycarbonate plastic (made from petroleum) against the coated glass—completing an electrical circuit. The computer is then able to figure out exactly where you touched the screen. Computer processors are made from silica wafers (more quartz) and also include a significant amount of copper and gold. Gold is used because it is a better conductor than copper and doesn’t tarnish the way silver or copper does. Most computers have nickel-metal-hydride (NiMH) batteries, which contain nickel, of course, along with cadmium, cobalt, manganese, aluminum, and the rare-earth elements lanthanum, cerium, neodymium, and praseodymium. The processor and other electronic components are secured to a circuit board, which is a thin layer of fibreglass sandwiched between copper sheets coated with small amounts of tin and lead. Various parts are put together with steel screws that are made of iron and molybdenum.
That’s not everything that goes into a tablet computer, but to make just those components we need a pure-silica sand deposit, a salt mine for sodium, a rock quarry for calcium, an oil well, a gas well, an aluminum mine, an iron mine, a manganese mine, a copper-molybdenum-gold mine, a cobalt-nickel mine, a rare-earth element and indium mine, and a source of energy to transport all of the materials, process them, put them together, and finally transport the computer to your house or the store where you bought it.
Practice Exercise 2.3 Where does it come from?

Look around you and find at least five objects (other than a computer or a phone) that have been made from materials that had to be mined, quarried, or extracted from an oil or gas well. Try to identify the materials involved, and think about where they might have come from. This pen (Figure 2.4.2) is just an example.
You may find it helpful to consult the overview of ore and industrial minerals, their uses, and notable Canadian sources provided in Appendix 4 to complete this exercise.
See Appendix 2 for Practice Exercise 2.3 answers.
Mining has always been a major part of Canada’s economy. Canada has some of the largest mining districts and deposits in the world, and for the past 150 years, we have been one of the world’s most important suppliers of metals. Extraction of Earth’s resources goes back a long way in Canada. For example, the First Nations of British Columbia extracted obsidian from volcanic regions for tools and traded it up and down the coast. In the 1850s, gold was discovered in central British Columbia, and in the 1890s, even more gold was discovered in the Klondike area of Yukon. These two events were critical to the early development of British Columbia, Yukon, and Alaska.
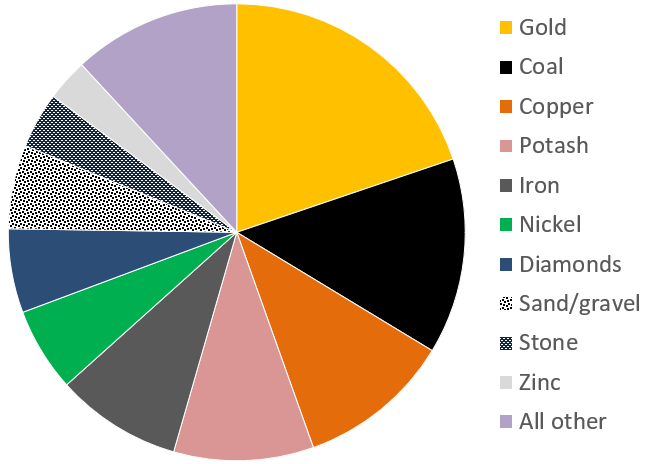
Canada’s mining sector had revenues in the order of $44 billion in 2017 (Figure 2.4.3). The 4 most valuable commodities were gold, coal, copper and potash, with important amounts from iron, nickel, diamonds, sand and gravel aggregates, stone and zinc. Revenues from the petroleum sector are significantly higher, at over $100 billion per year.
An ore deposit is a body of rock in which one or more metals have been concentrated to the point of being economically viable for recovery. Some background levels of important metals in average rocks are shown on Table 2.4, along with the typical grades necessary to make a viable deposit, and the corresponding concentration factors. Looking at copper, for example, we can see that while average rock has around 40 ppm (parts per million) of copper, a grade of around 10,000 ppm or 1% is necessary to make a viable copper deposit. In other words, copper ore has about 250 times as much copper as typical rock. For the other elements in the list, the concentration factors are much higher. For gold, it’s 2,000 times and for silver it’s around 10,000 times.
| Metal | Typical Background Level | Typical Economic Grade* | Concentration Factor |
|---|---|---|---|
| Copper | 40 ppm | 10,000 ppm (1%) | 250 times |
| Gold | 0.003 ppm | 6 ppm (0.006%) | 2,000 times |
| Lead | 10 ppm | 50,000 ppm (5%) | 5,000 times |
| Molybdenum | 1 ppm | 1,000 ppm (0.1%) | 1,000 times |
| Nickel | 25 ppm | 20,000 ppm (2%) | 800 times |
| Silver | 0.1 ppm | 1,000 ppm (0.1%) | 10,000 times |
| Uranium | 2 ppm | 10,000 ppm (1%) | 5,000 times |
| Zinc | 50 ppm | 50,000 ppm (5%) | 1,000 times |
| *It’s important to note that the economic viability of any deposit depends on a wide range of factors including its grade, size, shape, depth below the surface, and proximity to infrastructure, the current price of the metal, the labour and environmental regulations in the area, and many other factors. | |||
It is clear that some very significant concentration must take place to form a mineable deposit. This concentration may occur during the formation of the host rock, or after the rock forms, through a number of different types of processes. There is a very wide variety of ore-forming processes, and there are hundreds of types of mineral deposits.
Mining and mineral processing
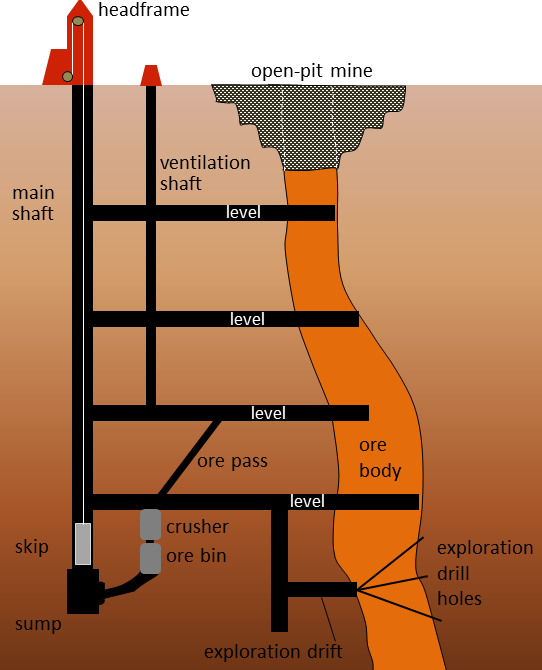
Metal deposits are mined in a variety of different ways depending on their depth, shape, size, and grade. Relatively large deposits that are quite close to the surface and somewhat regular in shape are mined using open-pit mine methods (Figure 2.4.4). Creating a giant hole in the ground is generally cheaper than making an underground mine, but it is also less precise, so it is necessary to mine a lot of waste rock along with the ore. Relatively deep deposits or those with elongated or irregular shapes are typically mined from underground with deep vertical shafts, declines (sloped tunnels), and levels (horizontal tunnels) (Figure 2.4.4). In this way, it is possible to focus the mining on the ore body itself. However, with relatively large ore bodies, it may be necessary to leave some pillars to hold up the roof.
In many cases, the near-surface part of an ore body is mined with an open pit, while the deeper parts are mined underground (Figure 2.4.4).
A typical metal deposit might contain a few percent of ore minerals (e.g., chalcopyrite or sphalerite), mixed with the minerals of the original rock (e.g., quartz or feldspar). Other sulphide minerals are commonly present within the ore, especially pyrite.
When ore is processed (typically very close to the mine), it is ground to a fine powder and the ore minerals are physically separated from the rest of the rock to make a concentrate. The rest of the rock is known as tailings. It comes out of the concentrator as a wet slurry and must be stored near the mine, in most cases, in a tailings pond.
The tailings pond at the Myra Falls Mine on Vancouver Island and the settling ponds for waste water from the concentrator are shown in Figure 2.4.5. The tailings are contained by an embankment. Also visible in the foreground is a pile of waste rock, which is non-ore rock that was mined in order to access the ore. Although this waste rock contains little or no ore minerals, at many mines it contains up to a few percent pyrite. The tailings and the waste rock at most mines are an environmental liability because they contain pyrite plus small amounts of ore minerals. When pyrite is exposed to oxygen and water, it generates sulphuric acid—also known as acid rock drainage (ARD). Acidity itself is a problem to the environment, but because the ore elements, such as copper or lead, are more soluble in acidic water than neutral water, ARD is also typically quite rich in metals, many of which are toxic.

Most mines have concentrators on site because it is relatively simple to separate ore minerals from non-ore minerals and this significantly reduces the costs and other implications of transportation. But separation of ore minerals is only the preliminary stage of metal refinement, for most metals the second stage involves separating the actual elements within the ore minerals. For example, the most common ore of copper is chalcopyrite (CuFeS2). The copper needs to be separated from the iron and sulphur to make copper metal and that involves complicated and very energy-intensive processes that are done at smelters or other types of refineries. Because of their cost and the economies of scale, there are far fewer refineries than there are mines.
There are several metal refineries (including smelters) in Canada; some examples are the aluminum refinery in Kitimat, B.C. (which uses ore from overseas); the lead-zinc smelter in Trail, B.C.; the nickel smelter at Thompson, Manitoba; numerous steel smelters in Ontario, along with several other refining operations for nickel, copper, zinc, and uranium; aluminum refineries in Quebec; and a lead smelter in New Brunswick.
Practice Exercise 2.4 Sources of important lighter metals
When we think of the manufacture of consumer products, plastics and the heavy metals (copper, iron, lead, zinc) easily come to mind, but we often forget about some of the lighter metals and non-metals that are important. Consider the following elements and determine their sources. Answers for all of these except magnesium are given above. See if you can figure out a likely mineral source of magnesium.
| Element | Source(s) |
|---|---|
| Silicon | |
| Calcium | |
| Sodium | |
| Potassium | |
| Magnesium |
See Appendix 2 for Practice Exercise 2.4 answers.
Image Descriptions
Figure 2.4.4 image description: An open-pit mine is dug to access the ore that is near the surface. For ore farther down, an underground mine will be constructed to access the ore. This diagram shows the main shaft (a large vertical tunnel) with four levels (horizontal tunnels) connected to it. The levels run from the main shaft into the ore body. A ventilation shaft runs up through the four levels in between the main shaft and the ore for air circulation.
Media Attributions
- Figure 2.4.1: “Ipad Air” © Zach Vega. Adapted by Steven Earle. CC BY-SA.
- Figure 2.4.2: “Ballpoint pen parts” by unknown. CC BY-SA.
- Figure 2.4.3: © Steven Earle. CC BY. Based on data from Natural Resources Canada.
- Figures 2.4.4, 2.4.5: © Steven Earle. CC BY.
a natural occurrence of rock or sediment that contains enough minerals with economically important elements that can be extracted at a profit
includes all materials extracted from the Earth except for fuels, metallic ores, water, and gemstones.
the typical level of an element in average rocks or sediments
a mine that is open to the surface
a vertical opening at a mine
in mining a decline is a sloped tunnel used to access lower parts of a mine with wheeled equipment
in mining, a horizontal mine opening
a product of ore processing that includes a specific ore mineral, separated from the rest of the rock
the fine-grained waste rock from a plant used to concentrate ore minerals
the production of acid from oxidation of sulphide minerals (especially pyrite) in either naturally or anthropogenically exposed rock
a refinery at which minerals are processed to produce pure metals

